Set to fade: is the brain doomed to degenerate?
Tuesday, Oct 11, 2011, 03:37 AM | Source: The Conversation
Malcolm Horne, Bradley Turner

Welcome to the sixth and final part of _On the Brain, a Conversation series by people whose job it is to know as much as there is to know about the body’s most complex organ. Here, Professor Malcolm Horne, deputy director of the Florey Neuroscience Institutes, and Bradley Turner, Senior Research Officer at Florey, examine why our brains degenerate, and ask whether there is any chance of changing this. Enjoy._
Why does age make the brain so susceptible to degeneration?
Dementia is a fatal disorder and the third leading cause of death in Australia – it increases markedly with age and approximately one in 100 Australians suffers from it.
Organs such as the liver, gut and blood repair and renovate themselves by regularly replacing damaged or ageing cells. Each of these organs has a nidus where stem cells manufacture these replacements. But brain and muscle cells endure with very little replacement.
While the liver can be almost completely repaired, even when as much as 50% is lost through injury or disease, there is only minimal repair of the brain following cell loss through damage or disease.
Animals with simple nervous systems and reptiles have brains that can self-repair but it appears that mammals lost this ability at some point in their evolution. We don’t know when or why this happened.
Going, gone
Brain cells can’t store their own energy but depend on being continuously supplied from blood and from their support cells, known as glia, which supply nutrients and other molecules needed for survival, and remove toxins. They act as the nerve cells’ private guardians.
Nerve cells have especially high energy demands, consuming about 20% of the body’s oxygen requirement and 25% of its sugar requirement, and brain cells die only a few minutes after blood supply fails.
Energy is needed to maintain a voltage across the membranes, which is necessary for producing the signals that pass between nerve cells. These connect to each other by long processes called axons or nerve fibres.
It’s worth remembering the axons going from brain to spinal cord or cord to muscle can be more than a metre long.
In nerve cells, as with all cells in the body, energy is also required for normal cellular functions. Cell organelles must be trafficked, cell parts repaired, renovated or extended, membranes repaired and synthesised.
Such processes demand the manufacture of proteins – DNA and RNA. In carrying out these tasks, molecules “wear out” and must be degraded and removed.
Also, a certain proportion of the newly-synthesised replacement proteins are defective and must be replaced.
Cleaning up
Nerve cells can be thought of as miniature factories requiring resources and expending energy for their survival, while producing waste by-products.
They require careful cleaning and maintenance, otherwise there’s the potential for protein to build up as “junk” within the cell, reducing the efficiency of the cell and increasing energy demands.
A common feature of the ageing brain is the appearance of abnormal protein junk piles in nerve cells. Quite often these are harmless, but in some cases they are signatures for degenerative brain disorders such as Alzheimer’s disease, Parkinson’s disease and motor neurone disease.
The exact reasons why these sticky protein clumps form in the brain is not well understood but the junk piles most likely reflect waste build up in nerve cells.
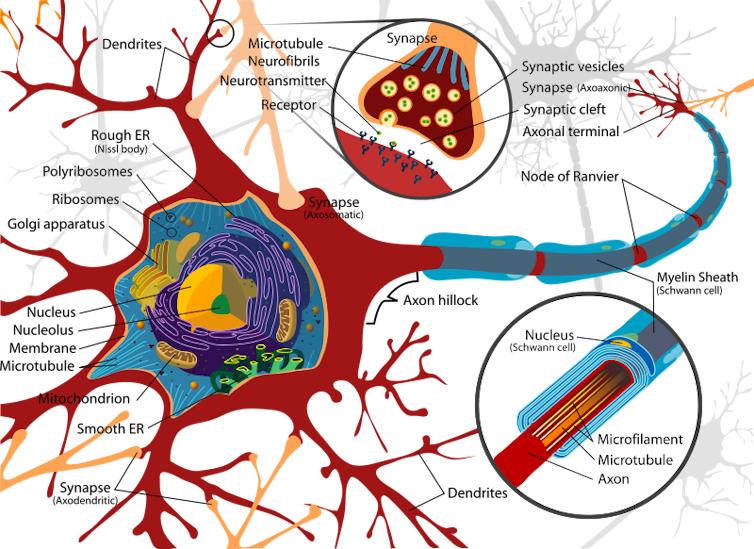
When junk accumulates in cells from other organs, those cells are marked for demolition and replaced. This option is not available for the brain, placing it at increased risk of degeneration with increasing age.
There is a suspicion, but no clear proof, that lifestyles with a focus on exercise, non-smoking, reduced fat and calorie intake, and continuing mental activity may reduce the risk of these degenerations.
There’s also a suspicion that, in some diseases such as Parkinsons, exposure to insecticides, which damage the efficiency of cell transport and disposal systems, may increase the risk of disease.
A key area of research is to better understand how and why these sticky protein clumps form in the brain.
When this is known, there will be a greater chance of designing drugs or lifestyles that prevent or slow the rate of brain decay.
Until then, we must continue to use our heads.
This is the sixth part of our series On the brain. To read the other instalments, follow the links below:
Part One: Picking your brains: what’s going on inside your head?
Part Two: Your brain knows the moves (you just get in the way)
Part Three: Brain’s addiction: what makes heavy drug users different?
Part Four: Brain’s addiction: is shooting up a disease or a choice?
Part Five: Rebuilding the damaged brain: can stem cells be used as repair kits?
Malcolm Horne receives funding from ARC, NHMRC Bethlehem Griffith Foundation, Comercialise Australia.
Bradley Turner receives funding from NHMRC, Bethlehem Griffiths Research Foundation and Motor Neuron Disease Research Institute of Australia.